Development of C—S Bond-Forming Reactions
My group is interested in the development of new and efficient methodologies to form C—S bonds. Various methods have been developed that describe the formation of C—S bonds; however, most of these processes require that the substrates be activated before any reactivity is achieved. I have developed a facile two-step process for the formation of C—S bonds starting from two alcohols, which was then applied to an intramolecular process for the formation of chiral tetrahydrothiophenes. As noted in the literature, the tetrahydrothiophene moiety is implicated in a plethora of biologically active molecules that possess hypocholesterolemic, anti-viral, anti-cancer, and anti-inflammatory properties. In an effort to expand the scope of the intramolecular C—S bond-forming process and to access other highly privileged, sulfur-containing scaffolds this chemistry shall be thoroughly investigated.
Synthetic Enzymes
My group is also interested in the development of synthetic enzymes that can catalyze an array of organic reactions. In my laboratory, the synthetic enzymes are made by imprinting a polymer with an organic molecule that resembles the transition state of the reaction to be catalyzed; these entities are fittingly termed molecularly imprinted polymers (MIPs).
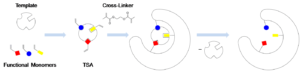
MIPs have been utilized in a plethora of sensor applications, as artificial antibodies and receptors, to perform chiral separations, in solid-phase extraction applications, and more recently as enzyme mimics in catalytic applications. Appealing aspects of MIPs as synthetic enzymes include: low cost, ease of synthesis, stability under extreme conditions (e.g.- low/high pH, high temperatures, organic/aqueous solvents), environmentally benign, recoverable, and high level of solubility for molecularly imprinted nanogels. Research is currently underway to access a shelf-stable synthetic enzyme that can be used to catalyze a well-known organic reaction.